Lucero joins SciLifeLab-coordinated nanoscale drug testing consortium
Biotech company, Lucero, joins the SciLifLab-coordinated Vinnova-backed Nanoscale drug testing consortium, alongside AstraZeneca, Pelago Bioscience, Karolinska Institutet and KTH. The consortium will now acquire new technology through Lucero, that will help solving challenges around miniaturization of the test systems and to make the implementation of these models cost- and labour-effective at scale.
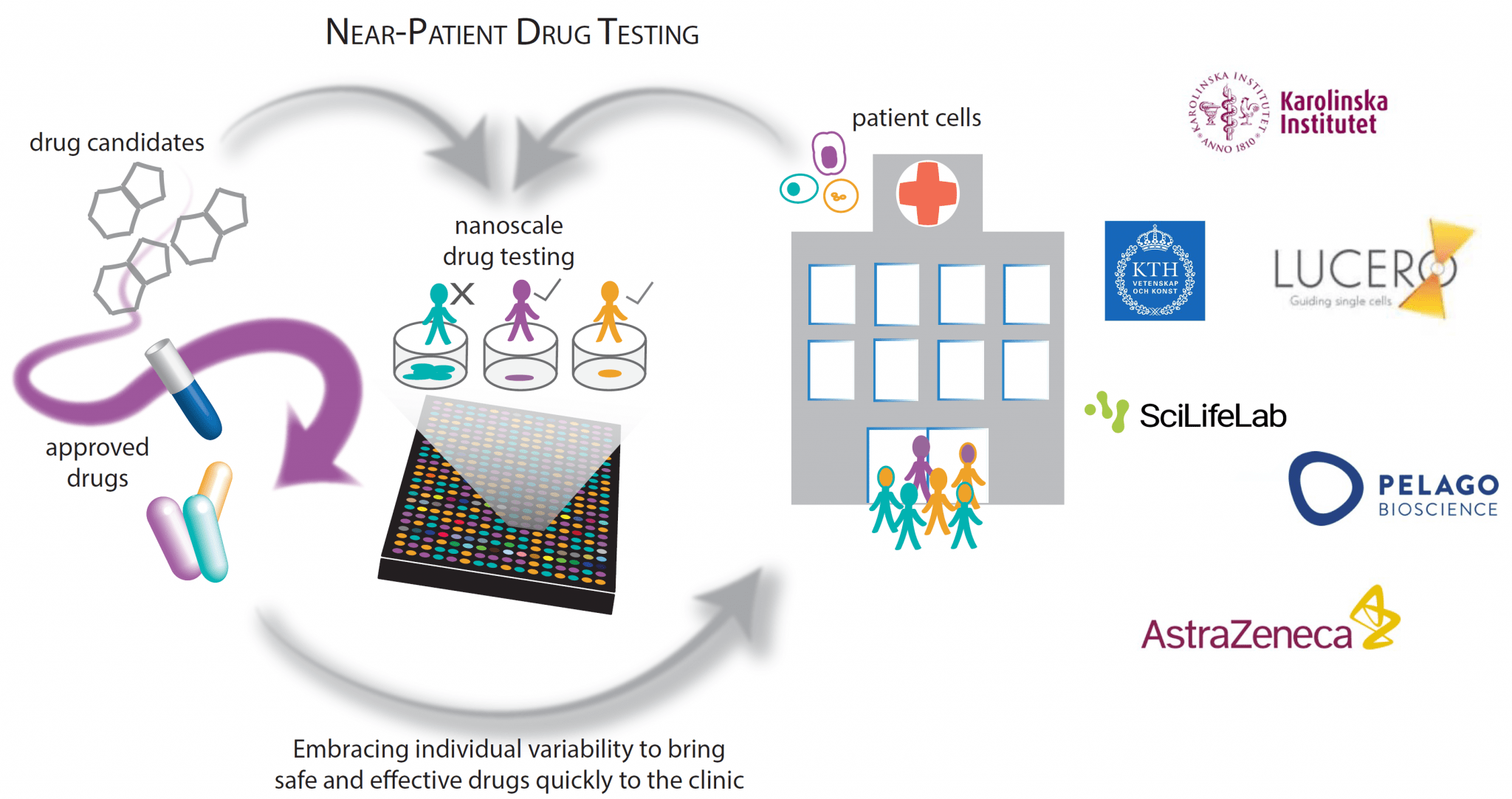
Accurately predicting efficacy and toxicity of emerging drugs in the clinic using conventional models of disease, particularly at the level of individual patients, can be very difficult. Optimally, cells should be collected directly from multiple patients (target and off-target tissues) with high disease representativity early on in the drug discovery process. However, there is currently no solution to cost-effectively embrace clinical heterogeneity, biological complexity and individual variability at the scale required for optimal prioritization in the drug development settings.
The nanoscale drug testing consortium aims to solve this by developing a “near-patient drug development”-strategy that includes using 3D cell models of patient-derived cells. The strategy aims to increase clinical success rates, enable the development of more effective treatments, and allow drug candidates to be matched with the optimal patient subgroup.
The consortium will approach this challenge by: 1) addressing the sourcing of relevant 3D cell models reflecting patient variability; 2) miniaturization of test systems for cost-effective testing; 3) multiparametric measurements to measure, for example, both safety and efficacy simultaneously.
Overall, the aim is to discriminate drug candidates that should not be pursued early on in the process, and create functional tests to monitor compound modulation of disease biology throughout the development process, eventually including individualized measures of both health and disease for precision medicine.
Positive results have already been achieved through initial tests and the consortium will now move on with its implementation.
Read more about the Nanoscale Drug Testing consortium.