New intermediate steps of gene expression characterized in human mitochondria
Researchers from SciLifeLab and Karolinska Institutet have managed to visualize the early steps on the initiation pathway of translation in human mitochondria by combining cryo-EM, fluorescence cross-correlation spectroscopy, and single-molecule imaging.
Translation of the mitochondrial proteins responsible for cellular respiration is carried out by distinct mitoribosomes with their own factors. How the different elements are assembled to initiate the translation was unknown.
In a recent study, co-led by SciLifeLab Fellow Alexey, researchers investigated mitoribosomal complexes in human mitochondria by using a structural biology approach.
To find out which specific states of the complexes lead to translation, intact complexes were extracted from the mitochondria. Using cryo-EM, postdoctoral researcher Yuzuru Itoh (SciLifeLab/SU) identified two previously unknown states, involving the small mitoribosomal subunit with initiation factors mtIF2 and mtIF3.
By applying computational single component analysis to the cryo-EM reconstruction, the researchers revealed an interdependence between mtIF3 and mitochondria-specific elements that prevents the premature association of the messenger and transfer RNAs.
Fluorescence cross-correlation spectroscopy and single-molecule imaging was used in order to confirm that the structures did represent a physiological state. The results illustrate that a mechanistic displacement of mtIF3 is essential for assembly of the complete initiation complex.
Based on these data, published in Nature Communications, the researchers could draw the conclusion that the newly identified states represent translation pre-initiation. No equivalent states have been reported in any other translation system, suggesting that this is a distinct feature of the human mitochondria.
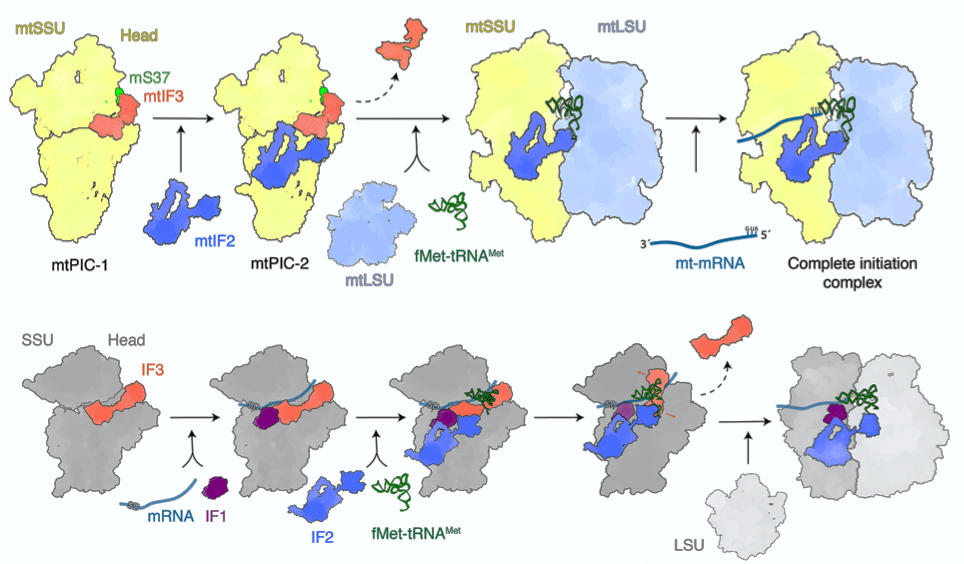
Schematic of translation initiation steps in human mitochondria (top) and bacteria (bottom). In the mitochondrial pathway mtIF3 and mS37 stabilize the small subunit head for accommodation of mtIF2. Then joining of the large subunit results in the conformational change of mtIF2 and GTPase activation that lead to the messenger RNA accommodation.





