New microscopy technique for effectively studying tumor spheroids
A new practical method for studying spheroids, organoids, and tumor-infiltrating immune cells at high resolution, has been developed by SciLifeLab researchers. This was achieved through expansion microscopy without losing the spatial organization. The article was published in Frontiers in Molecular Biosciences.
Cell cultures grown in three dimensions are better at mimicking the cellular environments and physiology of in vivo tissues, than two-dimensional cultures. Yet, the process of preparing samples with 3-D structures for light microscopy is often destructive, and using techniques such as physical sectioning might lead to the loss of three dimensional (spatial) information.
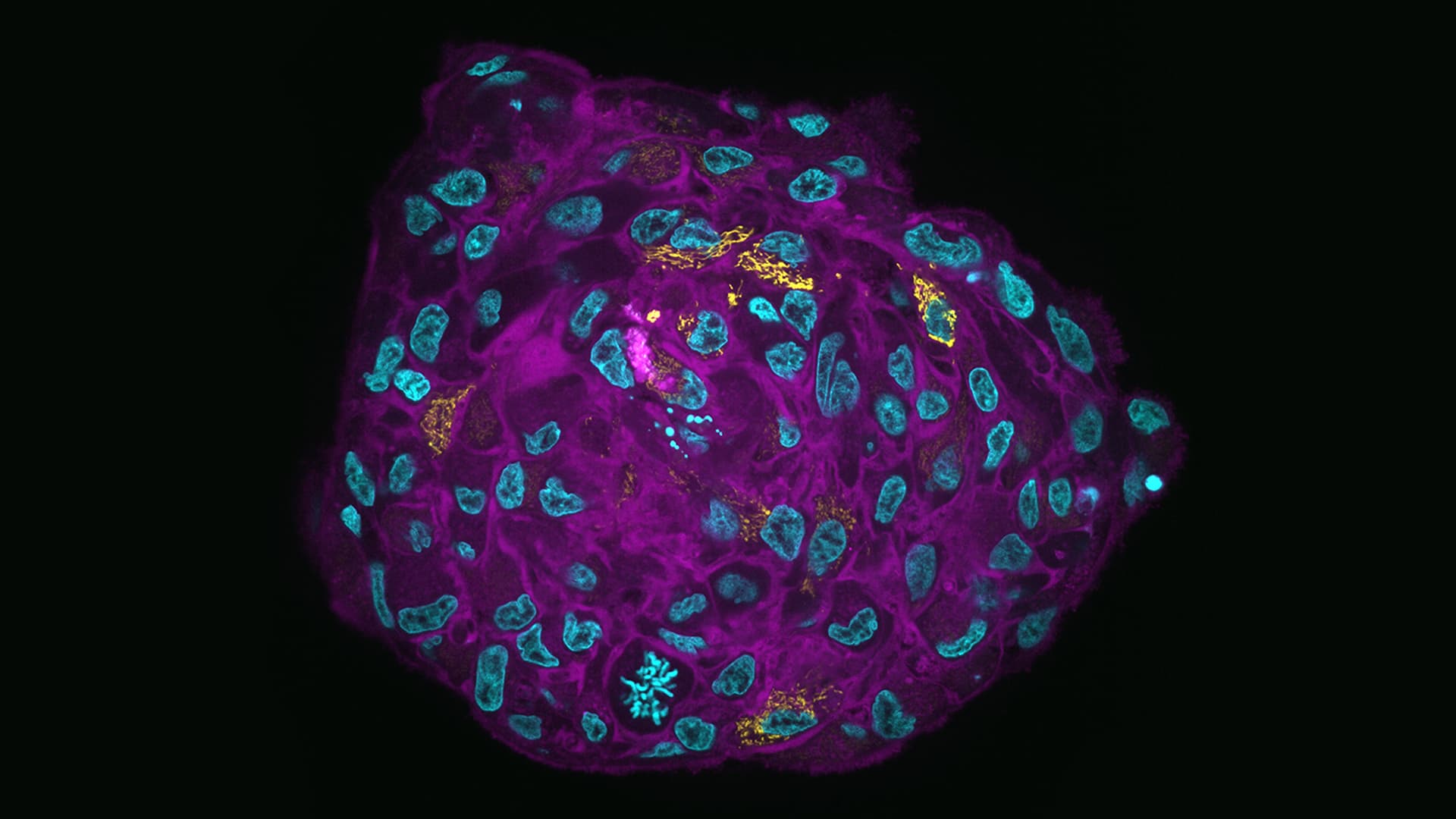
A research team, led by SciLifeLab researcher Hjalmar Brismar (KTH) and Steven Edwards (KTH), has developed a new practical method to tackle this issue. By fixing, anchoring, and embedding tumor spheroids in gel, it’s possible to produce a protein-hydrogel hybrid. By utilizing expansion microscopy they could produce highly transparent samples with high-resolution imaging over extended volumes, with significant reductions in light scatter and absorption. In using this methodology details were revealed in the center of organoids previously only observable with serial sectioning.
“Our goal with this study was to enable high-resolution microscopy of 3D cell cultures, such as tumor spheroids and organoids. We did this by optimizing the techniques for sample preparation, fluorescence labeling, light-sheet microscopy, and image analysis. We based much of the sample preparation on a novel method termed Expansion Microscopy which physically magnifies biological samples”, says first author Steven Edwards (SciLifeLab/KTH).
“One of the most unexpected difficulties was to keep track of the expanded samples as they became transparent during sample preparation, making them nearly invisible to the naked eye. But, this also made them fantastic for fluorescence microscopy”, says Steven Edwards.
Using spheroids is a great way to get a more accurate representation of the gradients of micronutrients found in tumor microenvironments. Flow cytometry is the standard method for quantifying protein expression, but as it requires dissociation of the spheroid into a single cell suspension, its microenvironment and spatial information is lost. This is something that the new technique might be able to change.
“The most important step in microscopy is sample preparation. No matter how good your microscope is, you cannot acquire high-quality images if you do not have a well-prepared sample. This study is a technological development that will allow us to investigate how immune cells interact with cancer cells growing in a 3D model of a solid tumor”, says Steven Edwards.
“In comparison to chemically cleared spheroids, the improved signal-to-background ratio of expanded samples greatly improved subsequent methods for image segmentation and analysis”.
Photo: Steven Edwards