New tool catalyze individualized precision cancer treatment in clinical trials.
A new tool, that helps molecular tumor boards to decide individual cancer treatments, has been developed by researchers from SciLifeLab and Karolinska Institutet, in collaboration with seven European comprehensive cancer centers. The portal has already been used to analyze tumor samples from more than 500 patients.
Today we have the ability to profile the molecular characteristics of tumors, which has opened the door to more effective therapies adapted to the condition of each patient. One of the biggest hurdles, however, is the volume and complexity of the molecular data, which medical teams need to interpret according to the latest scientific evidence in order to select the most appropriate treatment.
The process is time-consuming and often requires the use of a variety of databases and computational analytical tools, which are not easily accessible in a clinical setting. In addition, the number of molecular biomarkers grows every day, which generates an even bigger need for new technologies that can facilitate an accurate data interpretation and exchange of the generated knowledge.
To live up to these requirements, SciLifeLab researcher Janne Lehtiö (Karolinska Institutet) and his group, have developed the Molecular Tumor Board Portal, a clinical decision support system that combines a number of state-of-the-art methods to interpret the biological relevance of the genomic alterations observed in the patient’s tumor and prioritize available treatments accordingly.
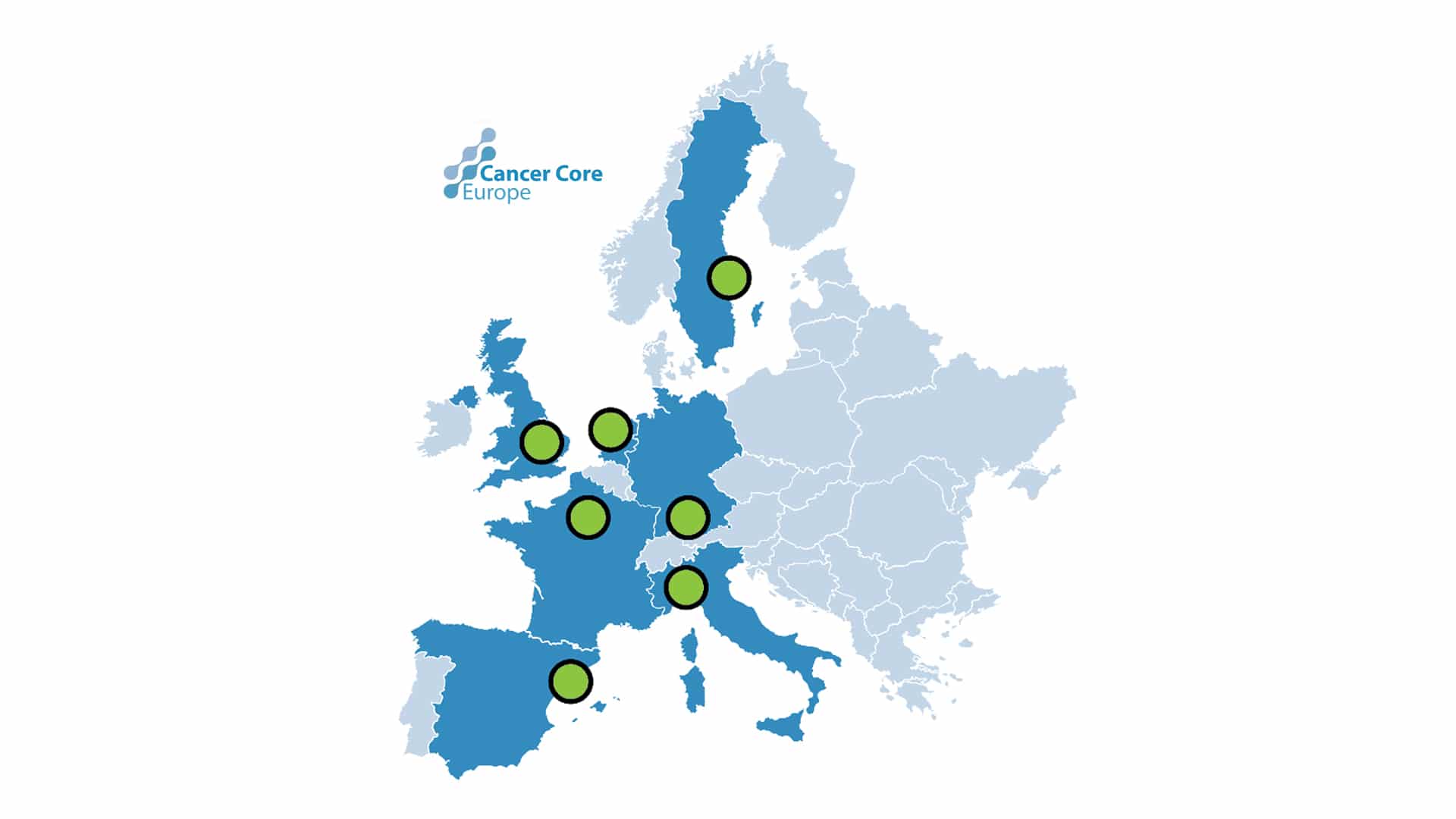
The portal is currently used by a collaborative network of seven European centers, which includes the Karolinska Comprehensive Cancer Center, to identify patients unresponsive to standard therapy but who might respond to novel drugs tested in clinical trials. The portal, which has been developed under the umbrella of the Cancer Core Europe, and ongoing work is described in an article published in Nature Medicine.
“The portal automates the tumor data interpretation, which eliminates potential errors due to manual processes and provides a systematic analysis based on expert consensus criteria”, says first author David Tamborero (SciLifeLab/Karolinska Institutet). “The use of this platform also avoids delays in the delivery of the results, which is critical for patients whose condition may rapidly deteriorate”.
Through a secure, web-based application, multidisciplinary representatives from each Cancer Core Europe center can share patient-centric reports and agree on clinical actions during weekly virtual meetings.
“From an oncologist view, the system has changed how we work. By using the portal in molecular tumor boards we have easy access to the latest knowledge on cancer mutations and can discuss the cases with colleagues from all the seven centers in a truly collaborative manner”, says Rodrigo Dienstmann, Principal Investigator of the Oncology Data Science Group at the Vall d’Hebron Institute of Oncology, Barcelona.
The researchers have observed that it takes approximately twenty-five reviewed patients to become familiar with use of the new system. After that, the discussion time for each case rarely exceeds a few minutes, something that is key when it comes to scaling the data-driven clinical decision process to a large number of patients.
An open-access version of the Molecular Tumor Board Portal genomics interpretation pipeline has been made publicly available at https://mtbp.org
“We believe that the use of the portal in a clinical setting is key to accelerate the translation of new research discoveries to the patients’ benefit”. “We are currently working to incorporate data from emerging biomarker discovery technologies such as proteomics and transcriptomics into the portal to further improve the selection of the most appropriate treatment for each patient”, says Janne Lehtiö.