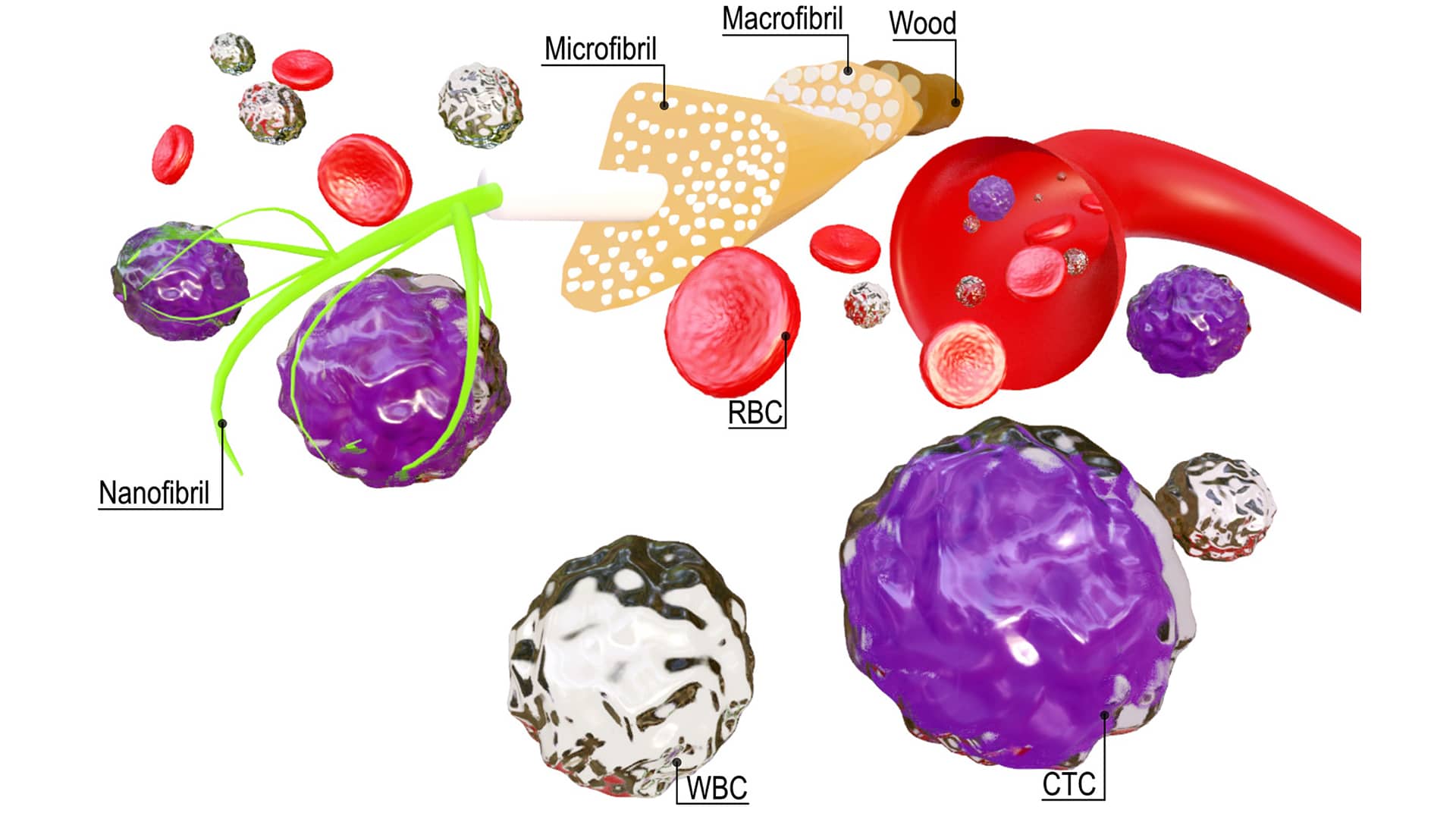
Promising diagnostic tool for cancer diagnostics
Researchers from SciLifeLab have developed a new cost effective cancer diagnostic method that is both fast and highly scalable. The new method is based on a novel multi-layered cellulose nanofibril coating and will be suitable for diagnostics in middle to low income countries.
According to reports from the World Health Organization (WHO), Cancer was responsible for 10 million deaths in 2018. A majority (65 percent) of those deaths occurred in low to middle income countries and these numbers are expected to grow even further (75 percent in 2030).
Healthcare and life expectancy have considerably improved in those areas during the past decades and soon the focus on infectious diseases and malnutrition will gradually shift towards cancer screening and management in the same way as for the rest of the world. For this reason, cost effective cancer diagnostic methods, without compromised sensitivity and specificity, is in high demand.
New methodologies, able to detect cancer cells in the bloodstream, have recently become a more popular alternative to invasive tissue biopsies and allow for early detection of metastasis, leading to a rapid treatment and subsequent follow-up.
Current technologies in the market, however, are rather complex, expensive and do not allow the release of viable tumor cells after isolation, which is a key feature needed for providing additional information about the cancer origin, aggressiveness and stage.
In a recent study, led by SciLifeLab researchers Aman Russom (KTH), Tharagan Kumar (KTH) and Ruben Soares (KTH), the researchers addressed these challenges by developing a novel multi-layered cellulose nanofibril coating functionalized with specific antibodies that targets circulating tumor cells. The method can be used for both capturing and releasing the tumor cells.
The method is highly scalable, cost-effective, simple, rapid and bio-based. It provides cell capture and cell viability figures as high as 97 percent. According to the researchers, the method, published in the journal Nanoscale, will soon be validated with samples from cancer patients to evaluate the potential clinical impact.