Research interests
The vascular endothelium has a key role in the maintenance of cardiovascular homeostasis. It produces a variety of active substances to maintain a non-thrombotic luminal surface, control vascular smooth muscle tone and regulate inflammatory processes. The structural integrity of the endothelium is crucial for normal vascular function and injury or inappropriate activation, termed ‘endothelial dysfunction’, is known to precede the development of both arterial and venous vascular disease. Endothelial dysfunction is an established response to exposure to risk factors, such as obesity and smoking, and can involve the expression of inflammatory chemokines, cytokines and leukocyte adhesion molecules, impaired availability of constitutively expressed cardio-protective substances, such as nitric oxide, and disruption in the regulation and resolution of coagulation.
Identification of endothelial-enriched genes
We developed a novel analysis method to identify endothelial-enriched genes (genes that are most highly expressed in this cell type, compared to others) through the deconvolution of unfractionated RNAseq data from multiple different human organs. Using this method, we published the first description of core human endothelial specific genes. As endothelial cells are known to differ significantly between tissue beds, with specialised functions depending on location, we have now expanded the analysis to investigate expression profiles in different organs. Using the human brain as a ´test case´ we showed a high level of consistency between our results and those from single cell RNAseq analysis, and the applicability of using this method to compare endothelial profiles in healthy and diseased tissue.
Investigation of uncharacterised endothelial cell gene function
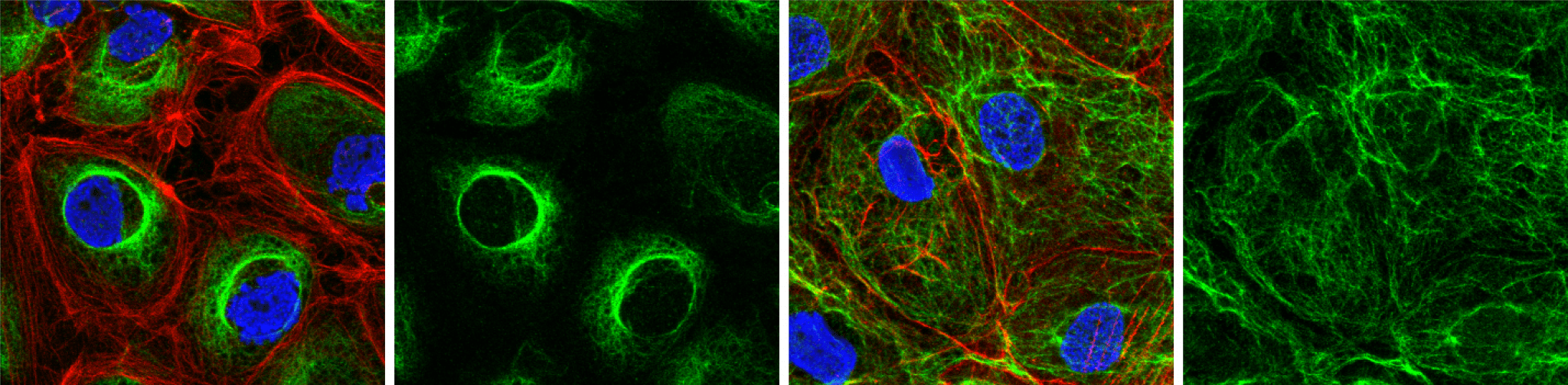
We study the expression of endothelial-enriched genes (see above) in population based or clinical cohorts, using both plasma proteomics and genetics, to identify candidates that are associated with endothelial dysfunction or cardiovascular disease development.
We prioritise functional studies of uncharacterised genes, which can be challenging to work on – often lacking tested reagents, such as antibodies. We use overexpression systems, knockdown and imaging techniques to characterise protein expression profiles and investigate their function using model vessels made from human endothelial cells. We can study how changes in the expression of these proteins can affect key endothelial functions, such as the control of leukocyte recruitment and thrombosis development from whole blood.
Endothelial response to inflammation
Endothelial cells are very important in the inflammatory response, controlling various processes including the expression of receptors and chemokines that induce the movement of immune cells from the blood into the tissue. Existing studies on how endothelial cells respond to inflammatory stimuli tend to lack a temporal aspect, with the focus on response induction, rather than secondary effects or resolution. Furthermore, there have been no descriptions of the influence of sex such dynamic EC responses, even though there are known to be sex differences in response to inflammation. Here, we study the transcriptional response of endothelial cells for extended periods following inflammatory cytokine exposure. Using bioinformatics analysis, we identify gene modules that represent early, mid and late response patterns. In addition to increasing our understanding of the endothelial inflammatory response, this data provides us with new candidates for functional studies, and potential plasma biomarkers for inflammation stage in patients. This is a collaborative project between our group and that of Dr. Marieke Kuijjer, UiO.
Plasma biomarkers for endothelial dysfunction
Currently, we lack clinical tools for the assessment of vascular health for risk profiling and monitoring of response following intervention to improve endothelial cell function. In this project, we used affinity plasma proteomics to measure levels of proteins that we previously identified as endothelial-enriched across human vascular beds (see above) in samples collected as part of the population-based study, the Swedish CArdioPulmonary bioImage Study (SCAPIS) pilot. We identified a panel of proteins which could be used to assess cardiovascular risk. Now we plan to expand this analysis to screen larger populations, and replication cohorts. This is a collaborative project between our group and that of Jacob Odeberg, KTH.
Plasma biomarkers for COVID-19 vascular pathology
Accumulating evidence indicates that endothelial dysfunction and associated prothrombotic state underlies severe COVID-19 pathophysiology. Why some individuals develop severe disease and who is at risk of long-term sequela remains an unanswered question. In a collaborative project, led by Jacob Odeberg, KTH, we aim to identify biomarkers associated with progression to severe or critical disease in patients initially presenting with moderate symptoms and biomarkers associated with long term complications. Candidate selection for screening include proteins that we previously identified as endothelial-enriched across human vascular beds (see above). Ultimately this information could be used to develop and validate biomarker-based prediction models as clinical tools to predict the likely course of disease progression and risk of short- and long-term complications.
Group members
- Philip Dusart, PhD, researcher
- Clément Naudin, PhD, Researcher
- Sofia Öling, PhD Student
Funding sources
- Swedish Research Council
- Swedish Heart Lung Foundation
- National CeRtre for Molecular Medicine, Norway
