Guiding future bladder cancer treatment with spatial proteomics
Researchers at KTH/SciLifeLab and Karolinska University Hospital develop new ways to improve treatment decisions for patients with advanced bladder cancer, using spatial proteomics to better predict who will benefit from novel immuno- and targeted therapies.
Bladder cancer is the ninth most common type of cancer, with over 600 000 new diagnosed patients each year globally. New treatments such as antibody-drug conjugates (ADCs) offer a more precise way to target cancer cells, but only roughly one out of three patients respond and it remains a clinical challenge to predict which patients will benefit from ADC treatment.
The study used the COMET platform and the multiomics workflow that was recently launched as a service at the Spatial Proteomics Unit at SciLifeLab, which enables multiplexed spatial analysis through sequential immunofluorescence combined with transcript detection using RNA Scope. By analysing multiple protein markers combined with RNA detection in the same cells, this method provides a detailed, spatially resolved view of tumour biology at single-cell level.
“The workflow we have optimized makes it possible to see both multiple RNA and protein targets within the same cells simultaneously. Targeting the RNA can be useful for protein markers that lack good antibodies, or as a way to validate single cell heterogeneity seen by protein expression. It also enables detection of other species such as bacteria or viruses within the human tissue microenvironment. Of relevance for this study of bladder cancer patients, the spatial characterization of various ADC therapeutic targets allows us to better predict which therapy is most likely to be effective in a certain tumour”, says Charlotte Stadler, Head of Unit of SciLifeLab’s Spatial Proteomics Platform.
By combining advanced molecular analysis with clinical expertise, the work is a clear example of SciLifeLab’s efforts in translating research into healthcare applications. The overall aim is to identify novel predictive biomarkers that can guide treatment decisions and improve patient outcomes.
“The prognosis for metastatic bladder cancer remains poor, therefore our goal with this integrated multi-omics approach is to identify the signals that show which patients are most likely to benefit from these novel therapies”, says Anders Ullén, professor of Clinical Oncology at Department of Oncology-Pathology, Karolinska Institutet and Senior consultant, Department of Pelvic Cancer, Theme CancerKarolinska University Hospital. “We also aim to determine the optimal treatment sequences for each individual patient as we move toward a precision‑medicine–based treatment paradigm guided by tumor characteristics.”
The research groups are now applying the spatial biology approach to evaluate how well the spatial patterns they observe correlate with clinical efficacy endpoints. The long-term goal is to support more informed clinical decision-making and enable more precise, individualized cancer care.
https://doi.org/10.1158/1538-7445.AM2025-5300
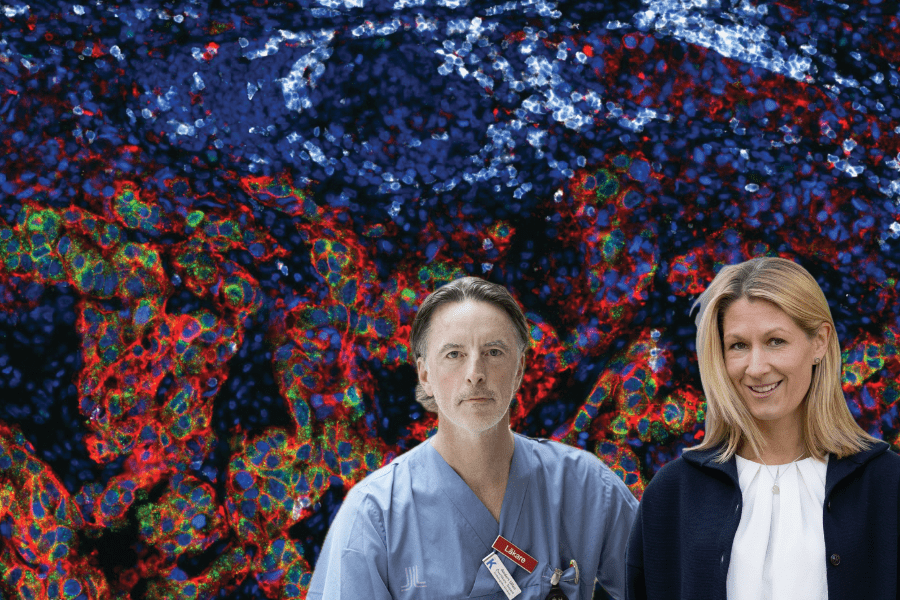
The collage was produced using the background image depicting a multiplexed immunofluorescence image acquired on the Lunaphore COMET platform to demonstrate the simultaneous detection of RNA and protein targets within a single tissue section of bladder cancer (photo credit: PhD student Pranauti Panshikar); a photo of Anders Ullén (photo credit: Håkan Flank/Radiumhemmets Forskningsfonder) and a photo of Charlotte Stadler (photo credit: Karin Nedler/SciLifeLab).